Supramolecular chemistry group deals with the following areas:
Bambusurils
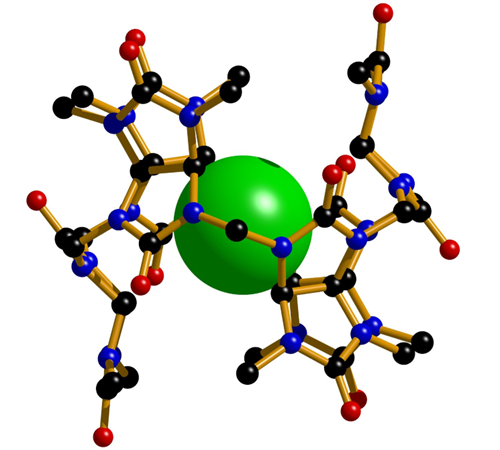
We are pioneers in the synthesis of bambus[n]urils (BU[n]) and in the investigation of their supramolecular properties. These macrocyclic compounds are based on glycoluril building blocks similarly to cucurbiturils. The unusual arrangement of glycoluril units provides bambusurils with the ability to act as anion receptors with high affinity and selectivity for various anions in both organic solvent mixtures and water. For instance, we showed that a bambusuril derivative recognizes and quantifies toxic perchlorate at 0.1 μM concentration in pure water.
Publications
Valkenier, H.; Akrawi, O.; Jurček, P.; Sleziaková, K.; Lízal, T.; Bartik, K.; Šindelář, V.: Fluorinated Bambusurils as Highly Effective and Selective Transmembrane Cl−/HCO3− Antiporters. Chem, 2019, 5, 429-444. link
Kandrnálová, M.; Kokan, Z.; Havel, V.; Nečas, M.; Šindelář, V.: Hypervalent Iodine Reversible Covalent Bond in Rotaxane Synthesis. Angewandte Chemie-International Edition, 2019, 58, 18182-18185. link
Yawer, M. A.; Havel, V.; Šindelář, V.: A Bambusuril Macrocycle that Binds Anions in Water with High Affinity and Selectivity. Angewandte Chemie-International Edition, 2015, 54, 276-279. link
Havel, V.; Yawer, M. A.; Šindelář, V.: Real-time analysis of multiple anion mixtures in aqueous media using a single receptor Chemical Communications, 2015, 51, 4666-4669. link
Havel, V.; Švec, J.; Wimmerová, M.; Dušek, M.; Pojarová, M.; Šindelář, V. : Bambus[n]urils: a New Family of Macrocyclic Anion Receptors. Organic Letters, 2011, 13, 4000–4003. link
Švec, J.; Nečas, M.; Šindelář, V.: Bambus[6]uril. Angewandte Chemie-International Edition 2010, 49, 2378-2381. link
Cucurbiturils
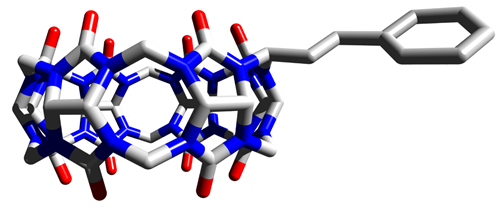
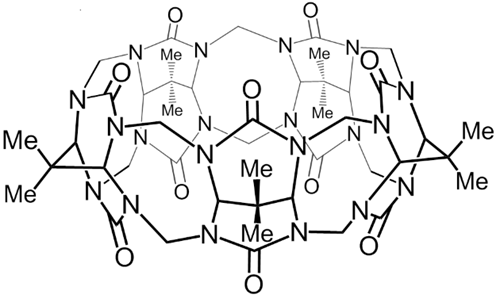
Cucurbit[n]urils (CB[n]) are water soluble macrocycles which are able to form highly stable supramolecular complexes with positively charged and neutral guests. We investigate the interaction of CB[n] with organic cations in order to design and synthesize molecular shuttles and swiches which can be driven by external input. Our effort to synthesis CB[n] derivatives resulted in first cucurbituril with a substituent attached solely to one methylene bridge. We also prepared CB[n] analog - pressocucurbituril.
Publications
Ustrnul, L.; Kulhánek P.; Lízal, T.; Šindelář, V.: Pressocucurbit[5]uril. Organic Letters, 2015, 17, 1022-1025 link
Gilberg, L.; Khan, M. S. A.; Enderesová, M.; Šindelář, V.: Cucurbiturils Substituted on the Methylene Bridge. Organic Letters, 2014, 16, 2446-2449 link
Koman, V.; Khan, M. S. A.; Babinský, M.; Marek, R.; Šindelář, V.: Supramolecular Shuttle Based on Inclusion Complex between Cucurbit[6]uril and Bispyridinium Ethylene. Organic Letters, 2011, 13, 6148-6151. link
Acyclic glycoluril oligomers
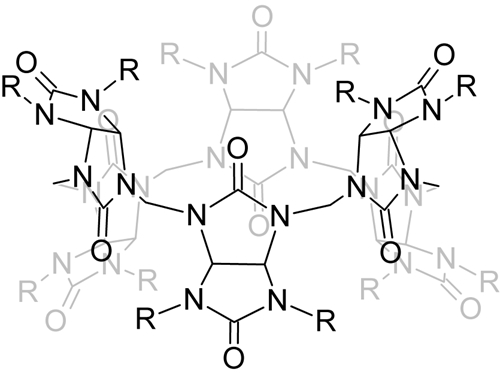
Cucurbit[n]urils (CBn) are generally prepared by polycondensation reaction between glycoluril and formaldehyde. However, this reaction results in limited number of products (n = 5 – 10) which separation is usually very difficult. In this project we investigate new synthetic approach, which allows the controlled preparation of new glycoluril oligomers and their transformation into macrocyclic structure. The ability of these compounds to behave as host molecules is also tested.
Publications
Štancl, M.; Gilberg, L.; Ustrnul, L.; Nečas, M.; Šindelář, V.: Synthesis and supramolecular properties of glycoluril tetramer. Supramolecular Chemistry, 2014, 26, 168-172 link
Štancl, M.; Hodáň, M.; Šindelář, V.: Glycoluril Trimers: Selective Synthesis and Supramolecular Properties. Organic Letters 2009, 11, 4184-4187. link